|
|
Pharmaceutical Solution Preparation System 500L with SS316L Mixing Tank (GMP Design)
Product Details:
Payment & Shipping Terms:
|
| Capacity: | 500L | Material: | SS316L |
|---|---|---|---|
| Application: | Pharmaceutical | Function: | Mixing, Storage |
| Control System: | PLC Automatic | Heating Method: | Optional |
| Surface Finish: | ≤0.8 μm | Cleaning Method: | CIP Optional |
| Voltage: | 380V | Certification: | GMP, ISO9001 |
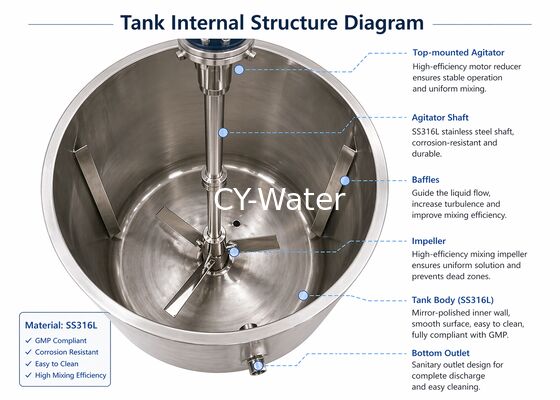
1. Product Overview
The 500L Pharmaceutical Solution Preparation System is designed for hygienic liquid preparation in GMP environments. Built with SS316L contact parts, it ensures corrosion resistance, easy cleaning, and stable mixing performance. The system is suitable for pharmaceutical, biotech, and laboratory-scale production where precision, hygiene, and repeatability are critical.
2. Applications
- Pharmaceutical liquid preparation (oral liquids, injections, syrups)
- Biotech and fermentation media preparation
- Cosmetics and personal care liquids
- Laboratory and pilot-scale production
3. Process Flow Diagram
Raw Water → Purified Water (PW/WFI) → Dosing → Mixing Tank (500L) → Heating / Cooling (Optional) → Filtration → Storage / Transfer
Process Description:
Purified water is introduced into the mixing tank, followed by controlled dosing of raw materials. The agitator ensures uniform mixing. Optional heating or cooling maintains process temperature. Final solution is filtered and transferred to storage or downstream processes.
![]()
4. Key Features
- SS316L hygienic design (product contact parts)
- Smooth internal surface (Ra ≤ 0.6 μm optional)
- Efficient mixing with uniform solution quality
- Optional heating/cooling jacket
- Optional CIP cleaning system
- Compact footprint for easy installation
- GMP-compliant design
5. Technical Parameters
| Item | Specification |
|---|---|
| Capacity | 500 Liters |
| Material (Contact Parts) | SS316L |
| External Material | SS304 |
| Surface Finish | ≤0.8 μm (≤0.6 μm optional) |
| Mixing Type | Top-mounted agitator |
| Mixing Speed | 0–60 rpm (adjustable) |
| Heating | Optional (electric or steam jacket) |
| Control System | Manual / PLC optional |
| Power Supply | 220V / 380V, 50/60Hz |
| Design Standard | GMP, FDA, WHO |
| Temperature Control | Optional |
6. Configuration List
| Component | Description |
|---|---|
| Mixing Tank | 500L SS316L tank |
| Agitator | Top-mounted mixing system |
| Manhole | Sanitary manhole |
| Level Indicator | Liquid level monitoring |
| Valves | Sanitary diaphragm valves |
| Piping | SS316L hygienic piping |
| Control Panel | Manual or PLC control |
| CIP System | Optional |
| Heating Jacket | Optional |
| Filter Unit | Optional |
7. Project Cases
- Southeast Asia Pharmaceutical Factory
Installed 500L solution preparation system for oral liquid production. Improved batch consistency and reduced contamination risk. - Middle East Biotech Company
Used for media preparation with optional heating system. Achieved stable temperature control and process efficiency.
8. Certifications & Standards
- GMP (Good Manufacturing Practice)
- FDA Compliance (design reference)
- WHO Guidelines
- ISO 9001 Quality Management System
9. FAQ
Q1: Can the system be customized?
Yes, capacity, material, control system and configuration can be customized based on project requirements.
Q2: Is CIP included?
CIP system is optional and can be integrated for automatic cleaning.
Q3: What material is used?
All product contact parts are SS316L stainless steel.
Q4: Can it be used for sterile production?
Yes, the system can be designed for sterile applications with proper configuration.
Q5: Do you provide installation support?
Yes, we provide global installation and commissioning services.
10. CTA (Call To Action)
Looking for a reliable pharmaceutical solution preparation system?
Contact us today for customized design, technical consultation and quotation.
Contact Person: Ms. Yanni.Wang
Tel: 86 15900488030
Fax: 86-21-66126659